The treatment of advanced Parkinson’s disease may be improved by a gel form of two common Parkinson’s medicines, levodopa-carbidopa, delivered through a tube inserted into the small intestine.
The study will be presented on April 25 as part of the Emerging Science program (formerly known as Late-Breaking Science) at the American Academy of Neurology’s 64th Annual Meeting in New Orleans April 21 to April 28, 2012.
The phase three randomized, double-blind, double-dummy clinical trial, showed that a levodopa-carbidopa intestinal gel (LCIG) (Duodopa, Abbott) had better results than levodopa-carbidopa immediate-release (IR) tablets. The study was supported by Abbott Laboratories, the company developing the new intestinal gel.
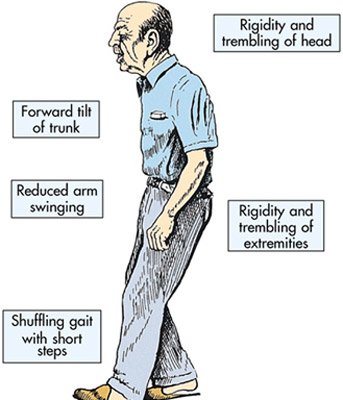
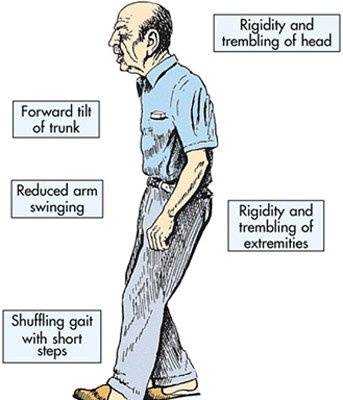
Parkinson’s disease is a brain degenerative disorder that affects around one million persons in the United States.
Most cases appear after the age of 50 and the symptoms include shaking, rigidity, slowness of movement and difficulty with walking in the early stages. In the advanced stages cognitive and behavioral problems may appear, including dementia. Other symptoms include sensory, sleep and emotional problems. Dopaminergic cells, brain cells that produce dopamine, a substance involved in controlling muscle movement, are slowly destroyed, and dopamine level drops dramatically.
Pills with levodopa-carbidopa, or dopaminergic medications, are used to replace dopamine. After a period of time, a substantial number of patients develop fluctuations in the response to dopaminergic medications. This will lead to shorter intervals during which symptoms remain adequately controlled, the effects of medication will start to “wear off‘ in between medication doses. Side effects include spontaneous and involuntary movements (dyskinesias) or “off” times. “Off-time” refers to periods of the day when the medication is not working well, causing worsening of Parkinson’s disease symptoms.
The study found that the continuous LCIG reduced “off-time” by an average of nearly two extra hours per day and improved “on-time” without troublesome movements by an average of two hours per day compared to people taking standard levodopa-carbidopa.
“Pills have a shorter half-life, but delivering the medication via gel allows for more continuous delivery and repairs the brain levels in a more normal way. “Off” time was reduced because the infusion of LCIG helps to deliver levodopa-carbidopa continuously, thereby avoiding the fluctuating levels that occur with standard oral levodopa-carbidopa therapy and that are thought to contribute to the development of wearing off,” said study author, Dr. C. Warren Olanow, Professor of Neurology and Neuroscience at the Mount Sinai School of Medicine in New York.
In the three-month double-blind trial, 71 patients were randomized to receive either the continuous infusion of LCIG and placebo pills or a placebo intestinal gel and pills that contained levodopa and carbidopa. At the start of the study, the average person had Parkinson’s disease for about 11 years and experienced 6.6 hours of “off-time” per day. A total of 93 percent of participants completed the study. The intestinal gel was infused through a portable pump connected to a tube implanted into the intestine (intrajejunal), similar to a feeding tube.
“It’s very similar to a feeding tube. The portable pump connected to the tube is discreet. It’s not like you’re running around with a big machine attached to you,” said Dr. C. Warren Olanow.
The results showed the intestinal gel offered significant improvements. Mean “off-time” decreased by 4.0 hours per day in patients receiving LCIG, an average of 1.91 fewer hours compared with the time in those taking the levodopa tablets. As well, mean “on-time” improved by 4.1 hours with LCIG, an average of 1.86 more hours compared with the time for the tablets. There was no significant change in “on-time” time with troublesome dyskinesia.
“Less “off-time” for people with Parkinson’s means more time during the day in which they can enjoy the benefits of levodopa-carbidopa therapy and experience improved quality of life,” said Dr. C. Warren Olanow.
The most common side effects associated with LCIG treatment involved complications due to inserting the device, abdominal pain, pain during the procedure and nausea.
Adverse events occurred in 35 (95%) patients receiving LCIG compared with 34 (100%) in the placebo group. The most common events were complications of device insertion (51%), abdominal pain (42%), procedural pain (32%), and nausea (25%). Other side effects included constipation (21%), orthostatic hypotension (18%), postoperative wound infection (17%), and incision site erythema (16%).
Treatment-related serious adverse events were reported in 5 patients (14%) in the LCIG group and in 7 patients (21%) in the levodopa-carbidopa IR tablets group.
Using LCIG procedure may avoid the need for brain surgery.
“We believe that benefits observed with this method of treatment compare favorably with other methods for treatment, such as deep brain stimulation, and avoid the need for an intracranial neurosurgical procedure,” said Dr. C. Warren Olanow.
Patients who have an anatomic impairment, are cognitively impaired, or prefer a gastrointestinal procedure over a brain operation are not good candidates for deep brain stimulation. In the same time, patients who don’t respond to levodopa are not good candidates for LCIG.
In Germany, LCIG, deep brain stimulation and apomorphine infusion, are the three treatment options used in Parkinson’s disease when conventional oral drug therapy fails to control motor fluctuations or dyskinesia, Dr. Jens Volkmann, Professor of neurology, University Clinic of Würzburg, Germany, told Medscape Medical News.
This study on levodopa-carbidopa intestinal gel was requested by the U.S. Food and Drug Administration to support an application for approval in the United States.